Oxygen: Element Properties and Uses
Oxygen is a vital element in the Earth's atmosphere, crucial for life processes and various industrial applications. It is widely used in medicine, manufacturing, and chemical processes.
Introduction to the Element
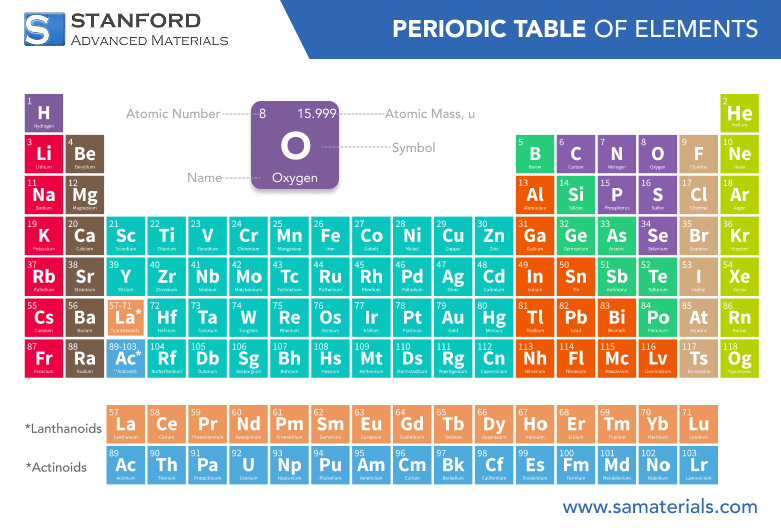
Oxygen is a chemical element with the symbol O and atomic number 8. It belongs to the chalcogen group of the periodic table and is classified as a nonmetal. In its most familiar form, oxygen exists as a diatomic gas (O₂) that constitutes about 21% of Earth's atmosphere, making it the second most abundant component after nitrogen. Oxygen is essential for aerobic respiration in most living organisms and is deeply involved in oxidation–reduction reactions that release energy in biological systems.
History and Naming
Although oxygen has existed on Earth for billions of years, it was not recognized as a distinct element until the late 1700s. In 1772, Swedish chemist Carl Wilhelm Scheele produced oxygen by heating mercury oxide and other compounds, though his findings were published later. Independently, in 1774, English scientist Joseph Priestley generated the same gas and described it as "dephlogisticated air," reflecting the prevailing phlogiston theory of combustion.
The true nature of oxygen was clarified by Antoine-Laurent Lavoisier, who recognized it as an element and demonstrated its central role in combustion and respiration. Lavoisier named the element "oxygen," derived from the Greek words oxys (acid) and genes (forming), because he believed oxygen was a universal component of acids. Although this assumption later proved incorrect, the name endured and remains one of the most recognizable in chemistry.
Occurrence and General Properties
Oxygen is extraordinarily abundant in nature. It is the most plentiful element in the Earth's crust by mass, largely in the form of oxides and silicates, and the dominant element in seawater when hydrogen is excluded. In the atmosphere, oxygen exists mainly as O₂, with a small fraction present as ozone (O₃) in the stratosphere, where it absorbs harmful ultraviolet radiation.
Chemically, oxygen is a highly reactive oxidizing agent. It readily accepts electrons from other elements, forming stable oxides and releasing large amounts of energy. This property underlies both controlled processes, such as respiration and industrial combustion, and uncontrolled ones, such as corrosion and fires.
Physically, oxygen is a colorless, odorless, tasteless gas at room temperature. When cooled below –183 °C, it condenses into a pale blue liquid, and at –218.8 °C it solidifies. Liquid oxygen is strongly paramagnetic, a property that allows it to be attracted by a magnetic field.
Commercial Production and Industrial Use
When large quantities of oxygen are required, the most common production method is the fractional distillation of liquid air. This process takes advantage of the different boiling points of air's major components. Because oxygen has a higher boiling point than nitrogen and argon, it is less volatile and can be separated efficiently.
The industrial process typically involves several steps: air is first filtered to remove dust and particles; moisture and carbon dioxide are absorbed using alkaline solutions; the purified air is then compressed and cooled to remove compression heat. Part of the compressed air is allowed to expand, lowering the temperature further until the mixture liquefies at approximately –196 °C. The liquid air is gradually warmed and distilled: lighter noble gases are removed first, nitrogen is separated next, and finally high-purity liquid oxygen is obtained. Repeated fractionation can yield oxygen with purities up to 99.5%, suitable for most industrial applications.
The steel industry is the largest consumer of pure oxygen. In basic oxygen furnaces, oxygen is blown through molten high-carbon iron to remove carbon and other impurities more rapidly and controllably than with ordinary air. Oxygen is also widely used in wastewater treatment, where it enhances biological decomposition, and in closed systems for waste incineration.
In aerospace engineering, liquid oxygen (LOX) serves as a powerful oxidizer in rocket propulsion systems. Its consumption depends on the scale and frequency of space missions. Pure oxygen is also vital in submarines and diving bells, where it supports life in sealed environments.
In the chemical industry, oxygen-enriched air or commercial oxygen has replaced ordinary air in the manufacture of acetylene, ethylene oxide, methanol, and other oxidation-controlled chemicals. In medicine, oxygen is indispensable in oxygen tents, inhalers, neonatal incubators, and anesthesia systems, sustaining patients during surgery and respiratory therapy. Oxygen further plays a crucial role in kiln-based industries such as glassmaking, cement production, and ceramics, where it improves combustion efficiency and temperature control.
Frequently Asked Questions
What does oxygen look like, and where is it found?
Oxygen is a colorless, odorless gas at room temperature. It is found in the atmosphere, dissolved in water, bound in minerals, and incorporated into nearly all living organisms.
Why is oxygen so important for living organisms?
Oxygen enables aerobic respiration, allowing cells to convert nutrients into usable energy in the form of ATP. Without oxygen, most complex life forms cannot survive.
How do plants and animals use oxygen?
Animals inhale oxygen to fuel respiration, while plants release oxygen as a by-product of photosynthesis and also consume oxygen during their own cellular respiration.
How is oxygen produced and used in industry and medicine?
Industrially, oxygen is mainly produced by fractional distillation of liquid air and used in steelmaking, chemical synthesis, water treatment, and rocket propulsion. Medically, it supports patients with breathing difficulties, sustains anesthesia, and maintains life in critical care environments.

 Bars
Bars
 Beads & Spheres
Beads & Spheres
 Bolts & Nuts
Bolts & Nuts
 Crucibles
Crucibles
 Discs
Discs
 Fibers & Fabrics
Fibers & Fabrics
 Films
Films
 Flake
Flake
 Foams
Foams
 Foil
Foil
 Granules
Granules
 Honeycombs
Honeycombs
 Ink
Ink
 Laminate
Laminate
 Lumps
Lumps
 Meshes
Meshes
 Metallised Film
Metallised Film
 Plate
Plate
 Powders
Powders
 Rod
Rod
 Sheets
Sheets
 Single Crystals
Single Crystals
 Sputtering Target
Sputtering Target
 Tubes
Tubes
 Washer
Washer
 Wires
Wires
 Converters & Calculators
Converters & Calculators




 Chin Trento
Chin Trento