Helium: Element Properties and Uses
Description
Helium is a noble gas that plays an essential role in various scientific and industrial applications. This blog post covers its properties, common uses, and preparation methods.
Introduction to the Element
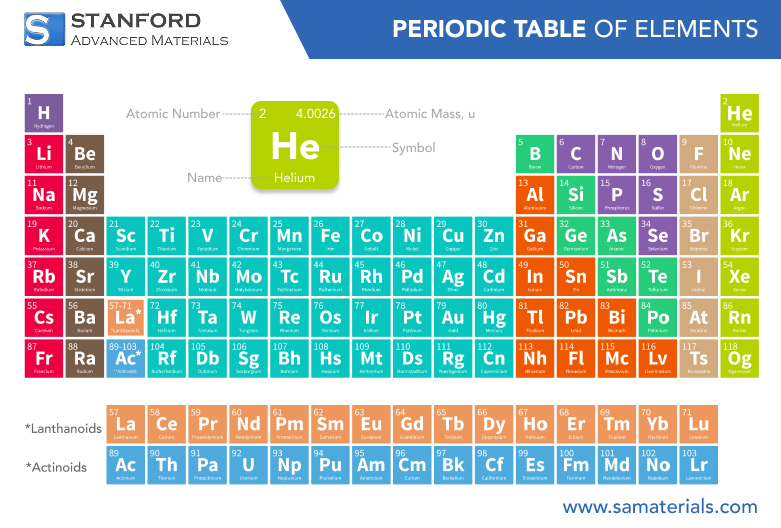
Helium is a colorless, odorless, tasteless, non-toxic, inert, monatomic gas that heads the noble gases group in the periodic table. It is the second lightest element in the universe, after hydrogen, and is primarily found in stars, including our sun. Helium is widely used in various fields due to its unique properties, including low boiling and melting points, which make it ideal for cryogenic and high-tech applications.
Chemical Properties Description
Helium is chemically inert due to its complete outer electron shell, making it extremely stable. As a noble gas, it does not readily form compounds with other elements under normal conditions. Its lack of reactivity makes it highly valuable in applications where chemical stability is crucial. Helium does not react with oxygen, nitrogen, or other gases at room temperature, and it remains non-flammable even in the presence of heat or fire.
Helium is also colorless and odorless, which adds to its safety and convenience in various industrial and scientific applications. Although it is mostly chemically inert, at high pressures and temperatures, it can form compounds with elements like sodium, potassium, and lithium under controlled laboratory conditions.
Physical Properties
Helium has several remarkable physical properties that make it useful in a variety of fields. Below is a table summarizing the physical properties of helium:
Property | Value |
Atomic Number | 2 |
Atomic Weight | 4.002602 u |
Boiling Point | -268.93°C |
Melting Point | -272.2°C |
Density | 0.1786 g/L (at 0°C and 1 atm) |
Color | Colorless |
Odor | Odorless |
Chemical Symbol | He |
Boiling Point (at 1 atm) | 4.22 K |
Specific Heat Capacity | 5.19 J/mol·K |
Helium's low boiling and melting points make it the only element that remains liquid at temperatures close to absolute zero, giving it unique applications in cryogenics and supercooling. For more information, please check Stanford Advanced Materials (SAM).
Common Uses
Helium has a wide range of uses across various industries, from medical applications to aerospace technologies. Some of the most common uses include:
- Cryogenics: Helium is crucial for cooling superconducting magnets used in MRI machines and in particle accelerators. Its low boiling point allows it to reach extremely low temperatures.
- Balloons and Airships: Helium is used to fill balloons and airships because it is lighter than air and non-flammable, unlike hydrogen, which is highly flammable.
- Breathing Mixtures: In scuba diving, helium is used in gas mixtures (like Trimix) to prevent nitrogen narcosis at high pressures.
- Leak Detection: Helium is used in the detection of leaks in high-vacuum systems, due to its small atomic size and low molecular weight.
- Welding: Helium serves as a shielding gas in arc welding because it prevents the oxidation of materials, ensuring high-quality welds.
Preparation Methods
Helium is primarily extracted from natural gas deposits where it accumulates in small quantities. The extraction process typically involves:
- Cryogenic Distillation: In this method, natural gas is cooled to very low temperatures, allowing helium to separate from other gases.
- Pressure Swing Adsorption: This method uses adsorbent materials to separate helium from other components in natural gas.
Helium can also be obtained as a by-product during the refining of uranium ore or from the radioactive decay of heavier elements like thorium and uranium, which emit helium as part of their decay process.
Related Industrial Products
Several products are dependent on helium, or are produced using helium in their manufacturing process. These include:
- Cryogenic Freezers: Used in medical and scientific applications, these systems rely on helium for cooling.
- Superconducting Magnets: Used in MRI machines and other high-tech equipment, these magnets require liquid helium to maintain low temperatures.
- Helium-Neon Lasers: These lasers, commonly used in scientific research and barcode scanners, depend on helium for their operation.
- Semiconductor Manufacturing: Helium is used in the production of semiconductors, especially for processes that require very low temperatures or controlled atmospheres.
Frequently Asked Questions
Why is helium used in balloons instead of hydrogen?
Helium is non-flammable, whereas hydrogen is highly flammable and poses a significant safety risk, especially in situations where an explosion could occur.
Can helium be made on Earth?
Helium is typically extracted from natural gas reserves, but it can also be produced by nuclear reactions, such as the decay of radioactive elements.
Is helium harmful to humans?
Helium is non-toxic and generally considered safe. However, inhaling large amounts of helium can displace oxygen in the lungs, leading to suffocation.
Why is helium so expensive?
Helium is relatively rare and expensive to extract, requiring specialized equipment for its separation and purification from natural gas.
How is helium used in medicine?
Helium is used in cryogenics to cool the superconducting magnets in MRI machines and is also part of breathing gas mixtures for patients with respiratory issues.

 Bars
Bars
 Beads & Spheres
Beads & Spheres
 Bolts & Nuts
Bolts & Nuts
 Crucibles
Crucibles
 Discs
Discs
 Fibers & Fabrics
Fibers & Fabrics
 Films
Films
 Flake
Flake
 Foams
Foams
 Foil
Foil
 Granules
Granules
 Honeycombs
Honeycombs
 Ink
Ink
 Laminate
Laminate
 Lumps
Lumps
 Meshes
Meshes
 Metallised Film
Metallised Film
 Plate
Plate
 Powders
Powders
 Rod
Rod
 Sheets
Sheets
 Single Crystals
Single Crystals
 Sputtering Target
Sputtering Target
 Tubes
Tubes
 Washer
Washer
 Wires
Wires
 Converters & Calculators
Converters & Calculators




 Chin Trento
Chin Trento