Astatine: Element Properties and Uses
Introduction to Astatine
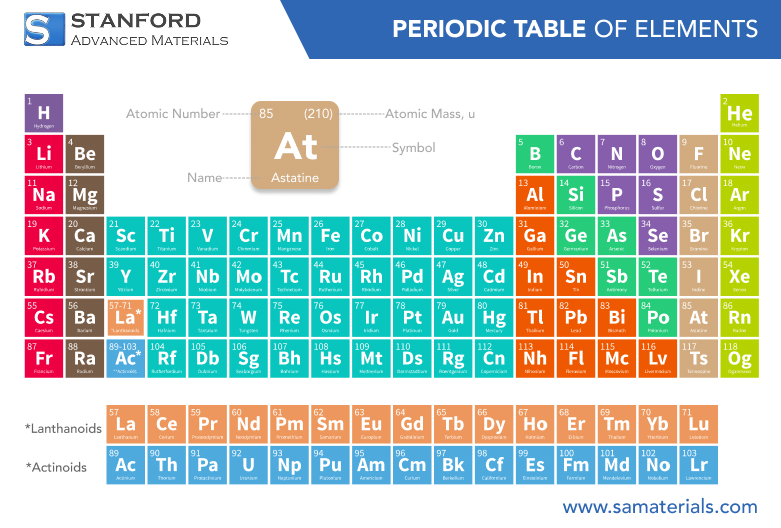
Astatine is a symbol At and atomic number 85 element that is among the naturally occurring and rarest elements on the planet. Astatine is among the halogen family of elements that possess some chemical resemblance with iodine and chlorine but is unique because it is highly radioactive and very rare. With a half-life of between hours and days depending on the isotope, astatine is not very long-lived in nature and therefore can be very challenging to research for scientists. Its most typical applications are confined to medicine alone, namely targeted alpha-particle therapy against cancer.
History and Naming
Astatine was first synthesized in 1940 by Kenneth Ross MacKenzie, Emilio Segrè, and Dale R. Corson of the University of California, Berkeley. They produced the element through bismuth-209 bombardment with alpha particles to obtain astatine-211, which is the most widely used isotope today in research.
The name "astatine" is derived from the Greek word astatos, meaning "unstable," in appreciation of its extremely radioactive and short-lived nature. Its discovery was the completion of the halogen group in the periodic table, although naturally occurring quantities are very tiny.
Physical Properties
Because of the rarity and radioactivity of astatine, some of its physical properties are instead calculated rather than measured directly. From theoretical calculations and extrapolation from other halogens:
Property | Value | Units |
Atomic Number | 85 | – |
Atomic Weight | ~210 | g/mol |
Electronegativity | ~2.2 | – |
~300 | K | |
Boiling Point | ~610 | K |
Density | ~7.0 | g/cm³ |
Astatine is expected to be a metalloid, having metallic and nonmetallic properties. Its color is assumed to be metallic and dark, possibly black or dark purple in bulk form, though no bulk samples have ever been observed.
Chemical Properties Description
Astatine resembles other halogens but its radioactivity is accompanied by unusual behavior. Some of the most significant chemical properties are:
• Oxidation states: Most characteristically −1, but in certain compounds +1, +3, +5, and +7 occur.
• Compound stability: Astatine compounds are generally unstable, with rapid decay due to the brief half-lives of its isotopes.
• Reactivity: It may form metal salts, e.g., astatides, but these very rarely survive for more than a few minutes.
• Electronegativity: At ~2.2, lower than that of iodine (~2.66), thus slightly less reactive in halogen chemistry.
For example, astatine can be theoretically caused to react with hydrogen and produce hydrogen astatide (HAt), just like hydrogen iodide (HI), but the compound quickly decomposes from radioactive decay.
Methods of Preparation
Because it has such low natural abundance and a short half-life, astatine is not produced from natural deposits. Astatine is produced artificially in nuclear reactors or cyclotrons. The most common method is:
1. Irradiation of bismuth-209 targets with alpha particles.
The astatine-211, with 7.2 hours half-life, is suitable for medical and experimental applications.
2. Chemical purification and isotopic separation: The astatine is separated quickly from the target by dry distillation or solvent extraction procedures to prepare it for laboratory experiments or clinical applications.
Due to these constraints, research and applications are time-critical and have to be carried out in highly specialized radiation-safe laboratories.
Applications
Despite its scarcity, astatine has promising applications in medicine and nuclear physics:
1. Targeted Alpha Therapy (TAT) for Cancer
Astatine-211 emits high-energy alpha particles that kill cancer cells while sparing much of the surrounding normal tissue. Clinical trials have tested thyroid cancer and brain tumors and discovered that astatine-labeled agents were able to deliver precise, targeted doses.
• Case Study: Recurrent ovarian cancer was treated with At-211 labeled antibodies in a 2015 trial conducted at University of Washington. The therapy was selectively cytotoxic to tumor cells and reduced systemic toxicity.
2. Radiopharmaceutical Research
Astatine isotopes find application in nuclear medicine as tracers to study metabolic pathways and develop new drug delivery systems. The short half-life offers high activity in small quantities, which is ideal for in vivo imaging and rapid action in treatment.
3. Nuclear Physics Experiments
Astatine's position in the periodic table also makes it well-suited for studying heavy element chemistry, isotope decay chains, and halogen chemistry under extreme conditions.
Conclusion
Astatine is an extraordinary element, marked by its rarity, radioactivity, and therapeutic potential. Though minute quantities occur naturally, artificial production has yielded new expectations for cancer therapies and advanced nuclear research. Its record—from discovery in 1940 to use today in radiopharmaceuticals—illustrates the unusual challenges and benefits of working with one of nature's most volatile elements.
Frequently Asked Questions
Why is astatine so rare?
It is extremely unstable and radioactive, decaying into other elements after hours or days, so only very small amounts exist naturally at any given time.
How is laboratory synthesis of astatine performed?
By irradiating bismuth-209 targets with alpha particles from nuclear reactors or cyclotrons to generate astatine isotopes, mainly At-211.
What are its major chemical properties?
Astatine is a halogen-like element but also produces radioactively unstable compounds. Its lower electronegativity and short-lived isotopes generate highly reactive and transient chemical species.
Why is astatine of interest for cancer therapy?
Its alpha radiation ensures malignant cells can be targeted specifically with little destruction of healthy tissue.
Does astatine have any industrial uses?
There are very little industrial applications; it is mainly used in medicine and nuclear science, unlike mass commercial processes.

 Bars
Bars
 Beads & Spheres
Beads & Spheres
 Bolts & Nuts
Bolts & Nuts
 Crucibles
Crucibles
 Discs
Discs
 Fibers & Fabrics
Fibers & Fabrics
 Films
Films
 Flake
Flake
 Foams
Foams
 Foil
Foil
 Granules
Granules
 Honeycombs
Honeycombs
 Ink
Ink
 Laminate
Laminate
 Lumps
Lumps
 Meshes
Meshes
 Metallised Film
Metallised Film
 Plate
Plate
 Powders
Powders
 Rod
Rod
 Sheets
Sheets
 Single Crystals
Single Crystals
 Sputtering Target
Sputtering Target
 Tubes
Tubes
 Washer
Washer
 Wires
Wires
 Converters & Calculators
Converters & Calculators




 Chin Trento
Chin Trento